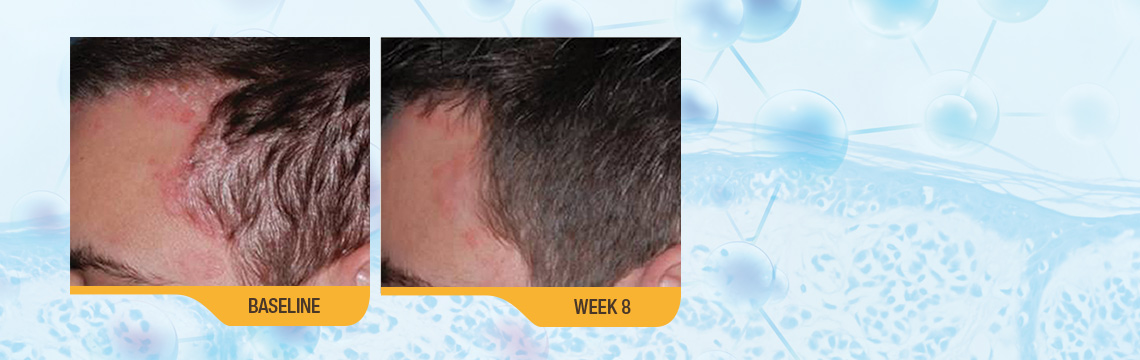
For your patients with scalp psoriasis
In a Phase III clinical study of moderate–severity scalp psoriasis, significantly more patients treated with SORILUX Foam achieved treatment success compared with vehicle foam. Treatment success was defined as an ISGA score of 0 (clear) or 1 (almost clear) at week 8 for scalp involvement.1
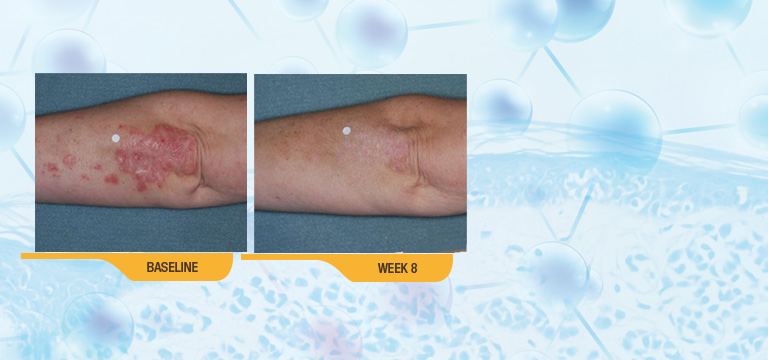
For your patients with body psoriasis
In two 8-week, Phase III studies, SORILUX Foam was shown to be safe and effective for treating mild to moderate plaque psoriasis of the body.2,3
Give your patients the only single-agent calcipotriene therapy available in an elegant foam formulation
SORILUX Foam delivers effective, steroid–free treatment for scalp and body psoriasis.
- Penetrates the skin barrier to deliver the molecule into the epidermis and dermis4
- Aqueous-based emulsion that is easy to apply to both hair-bearing and non–hair-bearing areas5
- Absorbs quickly without greasiness or residue4,5
- No ethanol, preservatives, or fragrances
- The contribution to efficacy of individual components of the vehicle has not been established
Most insured, eligible patients will pay $0 for a SORILUX Foam prescription.
References: 1. Feldman SR, Eastman WJ, Brundage T, Mills M. A multicenter, randomized, double-blind study of the efficacy and safety of calcipotriene foam, 0.005%, vs vehicle foam in the treatment of plaque-type psoriasis of the scalp. J Drugs Dermatol. 2013;12(3):300-306. 2. Feldman SR, Matheson R, Bruce S, et al; U0267-301 & 302 Study Investigators. Efficacy and safety of calcipotriene 0.005% foam for the treatment of plaque-type psoriasis: results of two multicenter, randomized, double-blind, vehicle-controlled, phase III clinical trials. Am J Clin Dermatol. 2012;13(4):261-271. 3. SORILUX Foam [package insert]. Raleigh, NC: Mayne Pharma LLC; 2019. 4. Data on file. Raleigh, NC; Mayne Pharma Commercial LLC. 5. Weiss S, Wyres M, Brundage T. A novel foam vehicle is consistently preferred by patients for dermatologic conditions. J Am Acad Dermatol. 2011;64(2):AB50.